As early as 1859 Mr. Plante showed that lead was the most favorable metal for use in secondary batteries, and he has since that time accumulated many proofs of this
superiority. Figs. 52 and 53 show the element as constructed to-day.
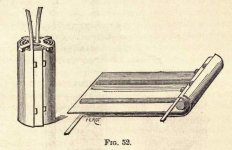

In a tall vessel made of glass, of rubber, or of ebonite are placed two sheets of lead rolled together parallel to each other, and kept apart by two strips of rubber rolled with them; these two sheets are immersed in a solution containing one tenth of sulphuric acid. The vessel is closed by a sealed stopper in which there is a hole through which the liquid is introduced and extracted, and through which the gases evolved during the charging may pass off. The apparatus is capped by an ebonite cover furnished with two clamp screws
which communicate with the two electrodes; there are also two clamps, which hold metallic wires to be heated and melted by the secondary current.
To charge this secondary element to its maximum, two of Bunsen's cells or three of Daniell's must be used. During the charging, one of the electrodes becomes oxidized, a brownish layer of peroxide of lead is soon seen, and the metallic aspect completely disappears; the other electrode only changes in appearance, its surface becoming covered with a grayish matter.
When it is charged to its maximum that is, when oxygen begins to free itself from the brown electrode it is well to separate the secondary cell from the active battery, as the polarizing current is no longer useful and is wasted.
The secondary element thus charged and left to itself can preserve a part of its charge several days, and at the end of a week it is still far from being exhausted.
The secondary cell when charged to its maximum has an electro-motive force equal to one and a half that of Bunsen's cell; it can bring to a glowing red heat a platinum wire large or small according to the dimensions of the cell, or, better, according to the size of its electrodes.
It can be easily understood, indeed, that the quantity of electricity furnished by the apparatus is in proportion to the extent of surface of the lead submitted to the action
of the polarizing current and covered with an active electro-chemical deposit.
It should be noted that the peculiar form of the electrodes offers a large surface and a small resistance under a small volume; so that one of Plante's secondary cells is equal to an active or ordinary cell of extraordinary dimensions; the small model has a surface of eight square decimetres, the large one a surface of four square decimetres.
The current furnished by a secondary element can produce chemical decompositions, act upon an electro-magnet, etc.; but if its intensity be measured in one way or another, with a galvanometer for instance, it is seen to diminish from the maximum of which we have spoken
above. This decrease is very slow if the circuit offers a great resistance, and if, as a consequence, there is a very small flow of electricity; it is on the contrary very rapid
if the circuit offers but a slight resistance, because the electricity flows in a large quantity.
A very curious and interesting fact is noticed during the discharge of the cell; it is apparently completely discharged, but if the circuit be left open several minutes, it has been ascertained that it recovers a certain energy and that it can still furnish a certain quantity of electricity.
The battery thus delivered of its first residue and left to itself for some time will furnish a second residue, less, of course, than the first. And this is not the last one, for several more can be obtained. Mr. Plante has very clearly explained this peculiarity. The secondary element, when it becomes active, discharges itself and at the same time polarizes, as all single-liquid batteries. This polarization attains in a certain time a force almost equal to
that of the already weakened secondary element, and the action ceases or is reduced to very little. If the battery then be left to rest, it depolarizes as do all single-liquid batteries polarized by their own action. As soon as the battery is depolarized it is again ready to furnish a current, but during this new discharge it again polarizes, and so on.
If we consider the secondary cell as completely or almost completely discharged, it may be recharged with two of Bunsen's elements, as in the first instance; but it is well to note that the more immediately after the discharge the new charge be given, the more rapidly it may
be given.
Moreover, the greater number of times a secondary element is charged and discharged, the better it is. In the beginning, when it is nearly new, there is an advantage in polarizing the electrodes, first in one direction and then in the other, and in reversing several times the
direction of the charge; but when the element is formed, great care must be taken to always charge it in the same direction. If this precaution be neglected it will take a much longer time to charge it, for the oxide of lead, which may still remain upon one of the electrodes, must be reduced and the previously negative plate oxidized. But after this operation the secondary cell will have recovered all its qualities: it may indeed be said to have gained some.
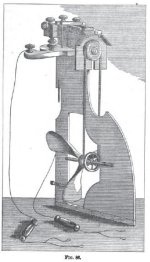
Fig. 54 shows a peculiar form given to the secondary element by Mr. Plante, and which he has called Saturn's tinder-box.

At the top are seen two clamps which hold a platinum wire stretched between them; each time that, by pressing with the finger, the two springs at the bottom are brought into contact the battery sends a current through the platinum wire, which is thereby brought to a glowing red heat, whence follows an almost instantaneous lighting of the candle. With one of these contrivances the candle may be lighted one hundred times, and it is only after these frequent lightings that it has to be recharged with three of Daniel's cells. That is a means 'of obtaining fire and a very economical means too, for the secondary element
spends nothing and the charging battery consumes but a few grammes of sulphate of copper for a prolonged working of the tinder-box.
This same apparatus can be used to touch off mines either in civil or military service; the experiment shows that with fine platinum-wire fuses (1/500 of an inch) combustion may be obtained through a copper wire 1000 yards long.
With a contrivance of this kind surgeons may cauterize a wound, and it has frequently been applied in that way. A secondary element is much more easily transported into a hospital or to the house of a sick person than active cells which it may replace.
Finally, secondary cells can be joined in quantity or in intensity and constitute batteries capable of producing all the effects of the most powerful ordinary batteries. Fig. 55 represents the secondary battery as disposed by Mr.Plante.
The number and dimensions of the cells can be varied according to the tension and quantity desired. Here there are twenty elements arranged in two rows.
At the top there is a very conveniently disposed commutator, which, in one position, joins the cells in quantity; in another position, at right angles with the first, it joins them in intensity. In the first position all the outer electrodes are joined to one metallic strip, and all the inner electrodes to another metallic strip, so that the whole arrangement represents a single cell with a large surface. It is in this condition that the charge is made; two of Bunsen's cells are sufficient, and they complete the charge in a longer or shorter time, according to the dimensions of the cells and to the extent of the surface of the lead to be polarized. In the second position the outer electrode of each cell is put into communication with the inner one of the following cell, and the apparatus becomes a real battery of twenty cells. It is in this condition that the battery is discharged, and it is equal, at first starting, to 30 of Bunsen's very large cells.
As the battery is being discharged the tension diminishes, as we explained when speaking of the single secondary element. If it takes one minute to charge the battery of secondary cells in quantity, it cannot be expected that the discharge of the cells in intensity will furnish the same effects as 30 of Bunsen's of the same size during a longer period than four seconds, for the apparatus furnishes no electricity and can only transform that which
has been given to it. Mr. Plante has made some exact experiments in this direction, and has found that in this transformation about one tenth is lost, or, in other words, the machine returns nine tenths of that which was given to it.
It is clearly seen that the secondary battery can only produce effects of very short duration, but in most cases this is all that is necessary.
If, for instance, a large number of mines are to be simultaneously exploded by means of fuses of fine wire, it may be done by placing all the fuses in divided circuits and by causing the current of the secondary battery to pass through them all at once. This manner of preceding is very economical, and it is certainly less laborious and costly to mount two of Bunsen's cells and to charge a secondary battery than to charge 20 or 30 of Bunsen's
elements, especially when the battery is only worked a few seconds and only four or five times during a day.