Meh...
Drive is taking forever to Zip and Download my data.
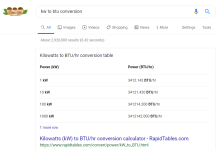
Heat Exchange Theory
1) Take a
liquid gas like Amonia
Any liquid can be heated to the point where it turns to gas
Any liquid, in enough vacuum, will evaporate into gas
Any phase change, so transition from liquid to gas, will have associated with it a big temperature change
P*V = n*r*T
Pressure x Volume ~= Temperature
Universal Gas Law
So.. does Amonia evaporate?
YES
This is why you use it to clean windows
Windex is Amonia
SO
Amonia is actually a gas that we trap as a liquid.
Example
You can capture Nitrogen Gas as a liquid and it will sit... you can pour it...
AND
We know that you can not really "Compress" liquids
RIGHT?
You compress Gasses and they convert to Liquids
SO
We correct the first statement to say that we "Compress Gas"
.
.
.
2) The Compressor increases the pressure and temperature of this gas by ... squishing it.
PV=T
P goes up, because we are squishing it
T goes up
We get this gas SUPER HOT
Much hotter than it was coming into the pump
We get this gas all compressed and angry... all the way until it is a liquid
(and this is how we choose an appropriate GAS (not liquid) for an AC Compressor)
... Same gas
Just hot and compressed now
So... Much more of it in the same volume
Saturated
When the gas is changed into a liquid it gets H O T
.
.
.
.
3) We then run this piping hot gas thru a "Heat Exchanger" which is nothing more than a bunch of copper pipes winding back and forth with a bunch of Aluminum fins... so a high surface area... with a Fan blowing air by... air which is much cooler than the gas which has been compressed into a liquid
View attachment 1
.
.
.
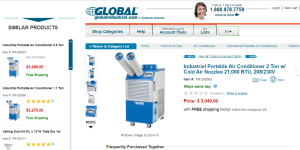
So the black tower in the pics is a "Compressor"
It is a pump
It converts gas to liquid
It makes this liquid HOT and under Pressure
The bottom of the compressor is where the gas comes in
Right where my finger is pointing.
... Some kind of mechanics happen inside of the Japanese High Reliability Pump
Out the top... comes angry hot liquid... unstable stuff...
it goes UP, OVER, DOWN, OVER, UP, and into the TOP of the radiator.
It then cycles thru the radiator
Top to bottom
Running around and around and around
4) When the compressed gas which is now a hot liquid exits the BOTTOM of the heat exchanger it is now MUCH COOLER.
All that blowing air over the fins has taken heat from our angry compressed gas
We have literally gone into the formula and MUCKED AROUND
PV=T
So... we forced T to drop
We held V, Volume, Constant
I am guessing P, remains constant, because behind us the pump is still pushing, pushing, pushing
If that is the case...
We now have an imbalance yes?
Obvoiusly P and V can not remain constant while T drops
So... Eh... I guess there is more to learn there
All I can tell you is that a bunch of heat has been blown off of our angry liquid.
This liquid which wants to return to equalibrium
(noodling here... noodling here... All things in life rest at equilibrium... only fools assume things taken out out equilibrium will rest easy*)
-methods